News & Topics


The Yamada Lab team placed third at the Sanken Football Tournament.

We held the ASPIRE Joint Symposium (IMPERIAL × Yamada lab).

Takumi Aoki, a second-year master's student (M2), received the Outstanding Student Presentation Award at the Electrochemical Society.

The Yamada Laboratory Fall/Winter Semester Open Lab for the 2025 Academic Year begins today. The schedule is as follows: October 6th and 7th during Period 1 and lunch break, and October 8th during lunch break and Period 4. Those wishing to observe should contact Professor Yasuyuki Kondo (yasuyuki.kondo@sanken.osaka-u.ac.jp) in advance. We look forward to the participation of all Environmental Energy students.

Junpei Kondo has graduated from the doctoral program.

We held a welcome party for international students.

Mr. Takeuchi (Graduate Students, M2 at that time) won the presentation award of the 92nd ECSJ Annual Meeting.
About us
Our research programs are centered on designing novel materials for various electrochemical reactions in energy storage, energy conversion, and electrosynthesis devices.
(1) We are developing high-energy-density and safe rechargeable batteries focusing on an electrode, electrolyte, and its interface. Through our unique strategy of controlling the coordination states of ions and solvent molecules in electrolyte materials, we are developing new functions and properties that are not found in conventional electrolytes. We are also pursuing the possibility of new rechargeable batteries by utilizing the developed electrolyte materials.
(2) With the energy crisis and global warming fast dawning upon us, there is a need for clean, renewable energy sources. Our research interest lies in developing the “Single Atom Catalysts (SACs)”-installed fuel cells with unprecedented atomic efficiency. Both experimental and computational tools are utilized to explore how SACs alter the reaction pathways and their reactivity.
(3) We are developing electrocatalytic technology to synthesize chemical fuels and feedstocks using discharged CO2, renewable electricity, and water. We perform fundamental studies of catalyst materials and integrate them into complete electrolyzer systems, including the engineering of catalysts, gas diffusion layers, and membrane electrode assemblies.
We will continue to explore unknown functions and properties of electrode and electrolyte materials and develop new technologies that solve energy and environmental issues.
RESEARCH
THEME
Research
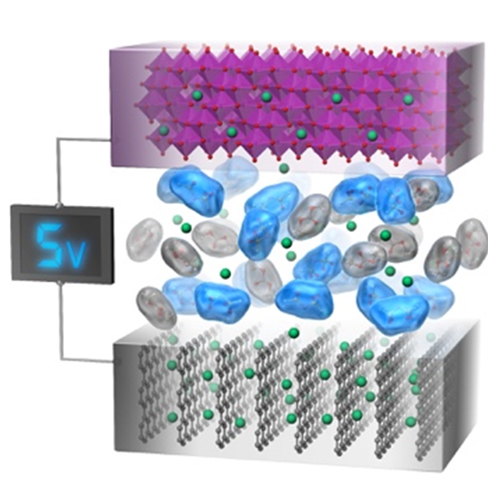 Next-Generation Rechargeable Battery Technology (Energy Storage)
Next-Generation Rechargeable Battery Technology (Energy Storage)
 Breakthrough Fuel Cell Technology (Energy Conversion)
Breakthrough Fuel Cell Technology (Energy Conversion)
 Innovative Power-to-X Technology (Electrosynthesis)
Innovative Power-to-X Technology (Electrosynthesis)
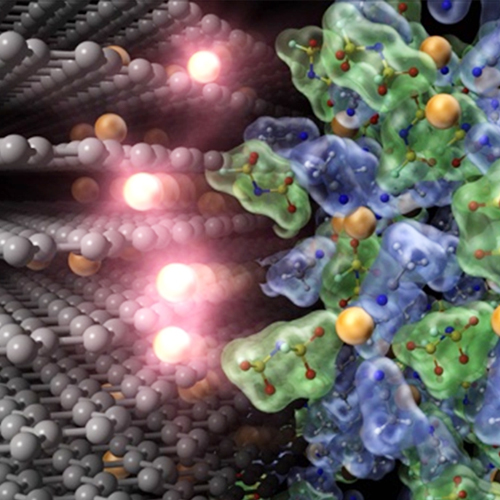 Pioneering “Advanced” Electrochemistry
Pioneering “Advanced” Electrochemistry